WHAT ARE CLINICAL STUDIES?
Clinical trials are research studies that test new treatments and interventions. They take place in carefully planned phases, each designed to answer specific scientific questions. Before they begin, they are thoroughly reviewed and approved, and they follow strict rules to ensure participant safety and confidentiality. Many studies also include healthy volunteers, meaning anyone may be able to take part and contribute.
Clinical trials are essential to advancing medicine and improving patient care. They help to:
HELP A CAUSE
Contribute to the development of new and better treatments.
HELP YOURSELF
Access innovative therapies at leading research centers.
HELP SCIENCE
Drive progress in science and patient care.
ONGOING TRIALS
Projects that aim to improve diagnosis and treatment for people with Parkinson’s disease by testing and
developing clinical biomarkers and novel treatments.

STRAT-PARK
STRAT-PARK aims to understand the heterogeneity of PD and identify subgroups of patients with distinct clinical and/or molecular characteristics, so that tailored experimental treatment can be developed and tested. The STRAT-PARK project will recruit PD and healthy people from Bergen, Trondheim and Canada. Healthy individuals are included to compare with patients and distinguish between Parkinson-related variation and naturally occurring variation in the population.

NADAPT
Progressive supranuclear palsy (PSP), Multiple system atrophy (MSA) and corticobasal syndrome (CBS) are severe neurodegenerative diseases that have apparent similarities to Parkinson’s disease (PD), but differ in key symptoms and prevalence. Results from PD studies show that nicotineamide riboside (NR) boost nicotineamide adenine dinucleotide (NAD) levels in the brain. The hypothesis for the NADAPT trial is that oral administration of NR can boost cellular NAD levels in the central nervous system of patients with PSP, MSA and CBS, rectifing metabolism and inhibiting neurodegeneration, which results in delayed disease progression and improvement of symptoms.
D-SPARK
D-Serine (DSR) is a naturally occurring amino acid that acts as a co-agonist at the N-methyl-D-aspartate receptor (NMDAR), a major glutamate receptor subtype, involved in synaptic plasticity. Growing evidence suggests that DSR may offer therapeutic benefits in neurodegenerative diseases. Previous studies indicate that D-serine is safe, well-tolerated, and a promising potential neuroprotective and disease-modifying treatment for Parkinson’s disease (PD). D-SPARK aims to assess whether orally administered D-Serine at a dose of 4000 mg per day (2x 2000 mg to ensure a stable bioavailability dosage throughout the day) can slow the neurodegenerative processes and lead to improvement in symptoms of people with PD.
ONGOING INITIATIVES,PREVENTION STUDIES AND BRAIN HEALTH STUDIES
At DECODE-PD, we are not only developing new treatments – we are working to prevent Parkinson’s disease altogether. Treating symptoms is progress, but stopping the disease before it starts is the ultimate goal. Thus, our group has studies that focus on prevention and brain health.
NADage
Aims to investigate the potential of nicotinamide riboside (NR) in reducing physical and cognitive decline in the frail population, while improving clinical symptoms of frailty.
ABOUT NADageABOUT FRAILTYNOR-RBD initiative
NOR-RBD is the world’s first preventive trials in Parkinson’s disease and will recruit individuals in the prodromal phase, identified via REM sleep behavior disorder (RBD).
ABOUT NOR-RBDABOUR RBDNADream
NADream aims to investigate the effects of NR on sleep quality in healthy adults. The hypothesis is that NR may improve the brain’s ability to “rest” during sleep, which may result in better sleep quality.
ABOUT NADreamABOUT SLEEP AND NRUPCOMING TRIALS
REVIVE-PD
The study aims to develop and test a novel regenerative treatment approach in which exosomes – microscopic extracellular vesicles derived from mesenchymal stem cells (MSC-sEVs) – are delivered intranasally. The goal is to reduce neuroinflammation, modulate oxidative stress and promote neuronal survival and regeneration, potentially slowing disease progression.
LEARN MOREPLATFORM TRIALS
SLEIPNIR is a pioneering platform designed to test multiple compounds for target engagement in parallel, advancing only the most promising candidates to phase III trials. HYDRA, our phase III platform trial, will investigate whether candidate treatments that pass testing at SLEIPNIR can slow or stop the progression of Parkinson’s disease.
LEARN MORECOMPLETED TRIALS
NOPARK
NOPARK recruited over 400 people with PD from all around Norway to investigate whether treatment with nicotinamide riboside (NR) protects nerve cells and delays disease progression. NR increases the level of an essential substance (NAD), which is beneficial for nerve cells’ energy turnover and survival. If the result is positive, the study could lead to new treatment that can slow down Parkinson’s disease.
learn more
N-DOSE
The NADPARK study (completed trial) showed significant biological and clinical effects with 1000 mg NR daily, but higher doses have not been explored in PD. So, it remains unknown whether improved responses can be achieved by escalating the dose. The objective of N-DOSE was to determine the Optimal Biological Dose (OBD) of NR in PD. The outcomes of this project will take us closer to developing NR into a PD-drug.
learn more
N-DOSE-AD
The NADPARK study showed significant biological and clinical effects with 1000 mg NR daily, but higher doses have not been explored neither in PD, nor in other neurodegenerative disorders, like Alzheimer’s disease (AD). The objective of N-DOSE-AD was to determine the Optimal Biological Dose (OBD) of NR in AD. The outcomes of this project will take us closer to developing NR into an AD-drug.
learn more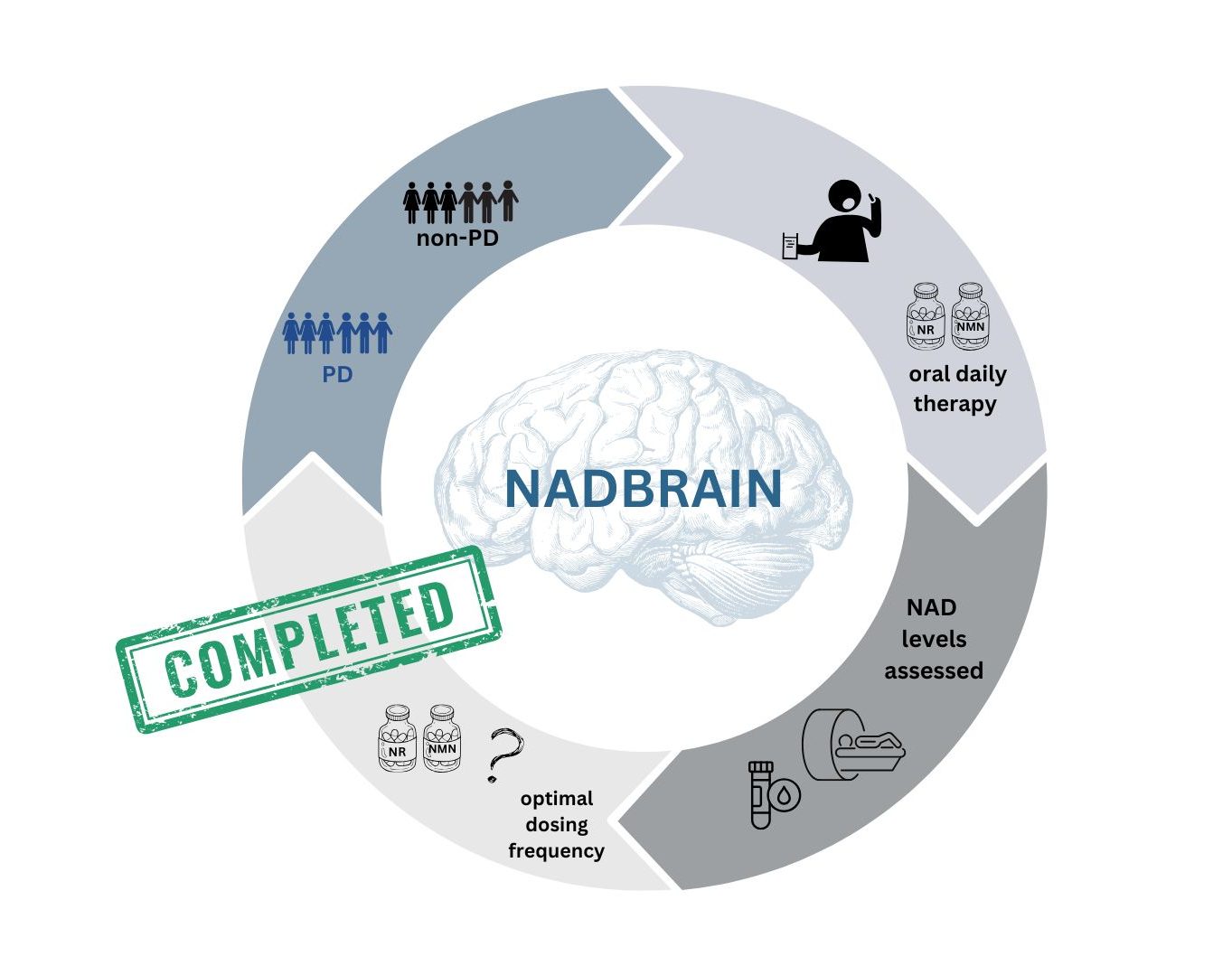
NADBRAIN
NADBRAIN is a pharmacokinetic study, that aimed to find out how much and how fast NAD levels increase in the blood and brain after taking NAD precursors for 8 or 28 days, and how long this increase lasts after the treatment ends. This is carried out using metabolite analyses in blood samples, and phosphorus-MR spectroscopy (31P-MRS) examinations of NAD levels in the brain, in both healthy individuals and individuals with Parkinson’s disease.
learn more
NR-SAFE
A phase I, randomized, double-blinded, safety trial of high-dose nicotinamide riboside (NR) in Parkinson’s Disease. This short trial took place during the first half of 2022 and has been completed showing that orally administered NR at a dose of 3000 mg daily is well tolerated in PD over a course of 4 weeks.
learn more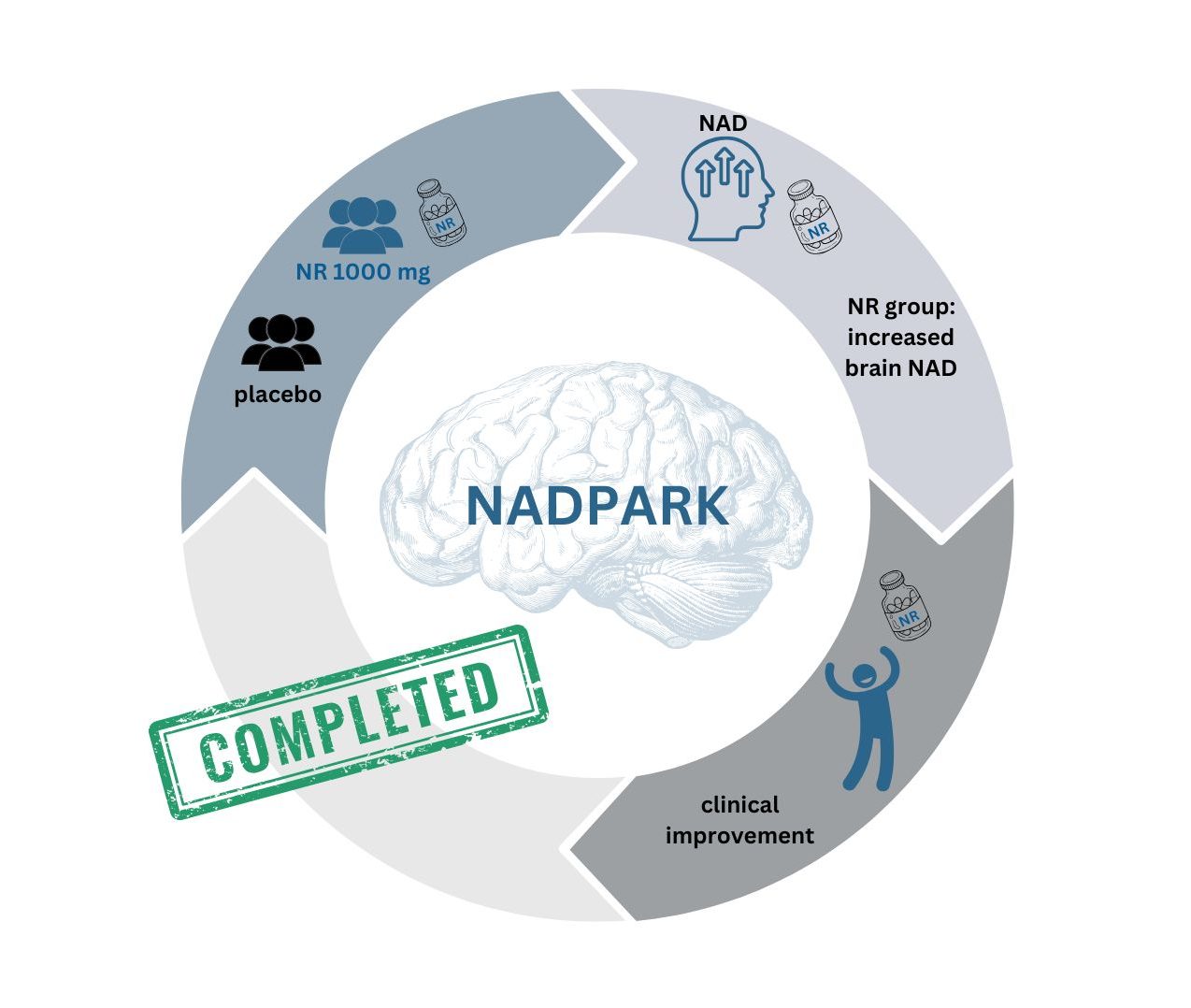
NADPARK
The NADPARK assessed tolerability, cerebral bioavailability and molecular effects of NR therapy in PD. The study showed encouraging results: intake of NR 1000 mg daily was well tolerated and led to a significant, but variable, increase in cerebral NAD levels and related metabolites in the cerebrospinal fluid (CSF). NR recipients showing increased brain NAD levels exhibited altered cerebral metabolism and this was associated with mild clinical improvement. NR augmented the NAD metabolome and induced transcriptional upregulation of processes related to mitochondrial, lysosomal, and proteasomal function in blood cells and/or skeletal muscle. Furthermore, NR decreased the levels of inflammatory cytokines in serum and cerebrospinal fluid.
learn more